Institutional News
Wang Xiaodong's laboratory found that the molecular mechanism of Estrogen-induced apoptosis and its physiological significance
Apoptosis is a programmed cell death pattern controlled by a series of genes that is activated during normal growth, development (a, metamorphosis from sputum to babies; b, enthalpy of insects; c, disappearance of sputum between higher mammals, such as embryonic development, embryos grow to week 5 and flat disc-shaped limb sprouts and the sputum between the fingers and toes disappears; d, cell death of the vitreous and lens of the eye, is an important step in the eye's light permeability), and certain physiological or pathological conditions. Apoptosis is essential even in mature individuals(For example, a, control of cell number and tissue size (average loss of 5-7 billion cells per adult per day); b, clearance of potentially dangerous cells by immune cells; c, conferring central nervous system plasticity). Apoptosis is characterized by cell blistering, shrinkage, nuclear fragmentation, chromatin condensation, chromosomal DNA fragmentation and global mRNA decay (Figure 1). The content of the final cell is encapsulated into apoptotic bodies by the cell membrane, and the apoptotic bodies are phagocytosed by nearby macrophages, so apoptosis does not cause an immune response. The body undergo apoptosis at all time. If the death pathway is blocked, the body will develop lesions. Some tumors are produced as a result of the inability of normal apoptotic cells to die.
Figure 1. Morphology of Apoptosis
According to the current research results, apoptosis can be divided into two pathways:
1.Extrinsic pathway which mediated by TNFα-TNFR (TNFα Receptor) or Fas-Fas ligand. For example, TNFα binds to the receptor TNFα Receptor on the cell membrane, thereby recruiting RIP1, and RIP1 further binds FADD and Caspase8 to form complex II (DISC, Death-Inducing Signaling Complex), which activates caspase8 and activates its downstream substrate Caspase3.
2.Intrinsic pathway (also known as the mitochondrial apoptotic pathway) is controlled by a class of proteins containing the BH (Bcl-2 homology) domain. The oligomerization of Bax and Bak at the outer model of mitochondria cause the outer mold of mitochondria to form pores, which release cytochrome C into the cytoplasm and bind to Apaf-1 and further recruit Caspase9 to form apoptotic complex in the presence of ATP/dATP. Apoptosome activates caspase9 by cleavage and further activates its downstream substrate Caspase3. In normal cells, oligomerization of Bax and Bak is inhibited by anti-apoptotic proteins Bcl-2, Bcl-XL and Mcl-1.
On August 13, 2019, Wang Xiaodong's laboratory published the Estrogen-Related Hormones Induce Apoptosis by Stabilizing Schlafen-12 Protein Turnover in《Molecular Cell》. The article systematically reveals the molecular mechanism and physiological significance of estrogen (E2) as endogenous inducer active intrinsic pathway in vivo.
In studying ovarian aging project, the researchers found that estrogen can induce HeLa cell death at 2.5-10 μM (above blood concentration) though apoptosis pathway. By knocking out the key proteins of the apoptotic pathway, caspase8 and caspase9, demonstrating estrogen-induced cell death through intrinsic pathway. Surprisingly, even blocking the intrinsic pathway does not block estrogen-induced cell death, but it can alter the morphology of cell death. Therefore, it can be speculated that there is a critical step in controlling the apoptotic pathway upstream of the mitochondrial apoptotic pathway. The authors then used compounds and CRISPR screening find that PDE3A (Phosphodiesterase 3) and SLFN12 (Schlafen family member 12) are key proteins in this apoptotic pathway. Knocking out any of the gene can completely block estrogen-induced apoptosis. It was subsequently confirmed that estrogen binds to the enzymatically active domain of PDE3A, suggesting that the conformation of PDE3A will change after binding to estrogen, and bind and stabilize its downstream substrate SLFN12. The authors subsequently found that the downstream substrate of the E2-PDE3A-SLFN12 complex was ribosome and SLFN12 binds to ribosomal RNA. E2-PDE3A-SLFN12 binds to the ribosome complex and blocks protein translation. It also blocks the binding of SRP (Signal Recognition Particle) to ribosomes. Because ribosomes require SRP to transported to the outside of the endoplasmic reticulum. The combination of E2-PDE3A-SLFN12 to ribosomes prevents the ribosome synthesized intracellular proteins, particularly intracellular mold proteins which will reduced protein levels of Bcl-2 and Mcl-1, making it unable to inhibit the activity of Bax and Bak. Bax and Bak oligomerization at the mitochondrial outer membrane forming pores release cytochrome C into the cytoplasm to activate Caspase9, and further activate Caspase3, eventually killing cells in the form of apoptosis (Figure 2). Since the E2-PDE3A-SLFN12 complex acts as a ribosome-inhibiting protein, even blocking the mitochondrial apoptotic pathway does not inhibit cell death. For the first time, the paper reveals the molecular mechanisms of the endogenous apoptosis-inducing agents-estrogen and biochemical pathways.
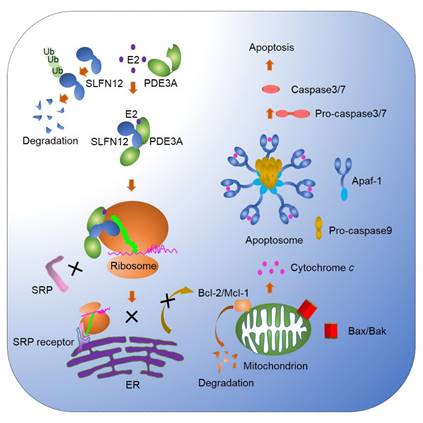
Figure 2. Model of estrogen (E2)-induced apoptosis
The subsequent question is what is the physiological significance of estrogen-induced apoptosis? The authors subsequently developed a monoclonal antibody against SLFN12 in cooperation with Abcam. Since SLFN12 can only be detected under estrogen-induced conditions, it can be used as an E2 induced apoptosis specific marker. According to previous research, estrogen can reach the concentration that induces apoptosis in one part of the body which is the placental tissue when pregnancy. The local concentration of estrogen in placental tissue can reach up to 12 μM, and according to previous literature reports, apoptosis is critical for successful implantation and placenta development. In order to detect whether estrogen-induced apoptosis is activated in human placenta tissue, Xiaodong Laboratory cooperates with Professor Zheng Qintian from Guangzhou Women and Children Medical Center. Guangzhou Women and Children Medical Center provides embryo samples of spontaneous abortion and from immunohistochemistry result, it was demonstrated that a strong SLFN12 signal was detected in the syncytiotrophoblast of human blastocytes, and the SLFN12 signal was co-localized with the activated Caspase3 signal. It is revealed that estrogen-induced apoptosis may plays an important role in human placental implantation and development. Whether this pathway affects the abortion of human embryos requires further research.
Dr. Dianrong Li (a postdoc at Wang’s laboratory), Dr. Chen Jie a Ph.D. student at Wang’s laboratory (joint graduate program with China Agricultural University) is the co-first author of this article. Dr. Youwei Ai (a postdoc at Wang’s laboratory) did important work. Professor Qintian Zheng from Guangzhou Women and Children Medical Center, Director Gu Xiaoqiong, Director Li Li, Dr. Di Che provided human endometrium and human embryonic tissues, and helped analyze the results of immunohistochemistry. Other authors of the paper include Dr. Zhaodi Jiang, Lin Li, Dr. She Chen, Dr. Huanwei Huang, Dr. Jiawen Wang, Dr. Tao Cai, Yang Cao, and Dr. Xiangbing Qi from NIBS. Xiaodong Wang is the corresponding author of this article. The work was supported by the Ministry of Science and Technology of China and the Beijing Municipal Science and Technology Commission.
Original link: https://www.cell.com/molecular-cell/fulltext/S1097-2765(19)30497-6



