Institutional News
Science Translational Medicine| A Study from Dr. Erquan Zhang's laboratory discovers a natural product capable of reversing the circadian phase through an RUVBL2-directed mechanism
On May 6, 2020 ---- Dr. Erquan Zhang laboratory published a new research article titled “Chemical perturbations reveal that RUVBL2 regulates the circadian phase in mammals” online in Science Translational Medicine. Using a high-throughput chemical screen with stringent criteria, the authors discovered a natural product (cordycepin) that is capable of causing very large phase shifts of mammalian circadian clock; cordycepin can thus be understood as a potential drug for the treatment of diseases related to circadian phase disorders like jetlag. Mechanistically, the triphosphate metabolite of cordycepin reorganizes the composition of the mammalian TTFL oscillator to elicit its phase-shifting effects.
Circadian rhythms are manifest in most physiological processes in mammals, including most obviously in sleep-wake cycles, heart rate, metabolism, and hormone secretion. These rhythms are driven by endogenous circadian clocks. The molecular mechanism of circadian rhythms is based on a transcriptional-translational feedback loop, which was awarded with the Nobel Prize in Physiology or Medicine in 2017. There are three parameters to describe circadian rhythms: period, amplitude, and phase. Most of the previous investigations of circadian oscillation have focused on the clock period, because it has been a fundamental question in the field to understand how circadian rhythms are gated at around 24 hours. Substantially fewer studies have examined the phase and amplitude of the clock, which are very sensitive to environmental perturbations. However, the circadian phase turns out to be the most related to human health in modern society, as for example in circadian rhythm sleep disorders that result from jet leg and shift work, sleep phase advance disorder in aged people, and metabolic diseases resulting from circadian phase dysregulation. Importantly, 75% of FDA-approved clinical trials are with agents that show dosing-time-dependent efficacy or toxicity, based on the analysis of an ~50 year history of more than 100 clinical trials. A better understanding of circadian phase regulation should therefore provide recommendations for future chronotherapies.
To identify small molecules that regulate the mammalian clock phase, the authors screened an institutional pilot chemical library comprising ~10,000 National Institutes of Health (NIH) clinical/preclinical trial drugs, purified natural product compounds, and synthetic compounds. Fortunately, the natural product cordycepin was discovered to generate a robust and reliable “antiphasic” phenotypes at the human cell, mouse tissue, and whole-animal behavior levels. Notably, this is the first small molecule discovered to date that can penetrate the blood brain barrier (BBB) and re-set the phase of circadian central pacemaker (SCN). Interestingly, cordycepin could also enhance the amplitude of the circadian rhythm when administrated at the trough of Per2 gene expression; the application of cordycepin for diseases related to weakened circadian clock are now being investigated (Fig. 1).
Structurally, cordycepin is an adenosine analog. In further SAR analysis, the authors tested 117 structurally related nucleosides and found three additional adenosine derivatives that caused an antiphasic phenotype. Despite this apparent specificity of adenosine derivatives in the observed phase shift, the authors establish that the phase shifting effect of cordycepin results from intracellular activity via adenosine transporters, rather than through adenosine receptor signaling activity. It is also notable that this phase-shift phenotype appeared to be unrelated to other known signals that affect the clock phase (cAMP, Ca2+ pathways) or transcription in general. Intraperitoneal injection of cordycepin caused rapid induction of E-box genes within 1 hour of cordycepin treatment, indicating a novel mechanism gating the circadian phase at the transcriptional level (Fig. 1).
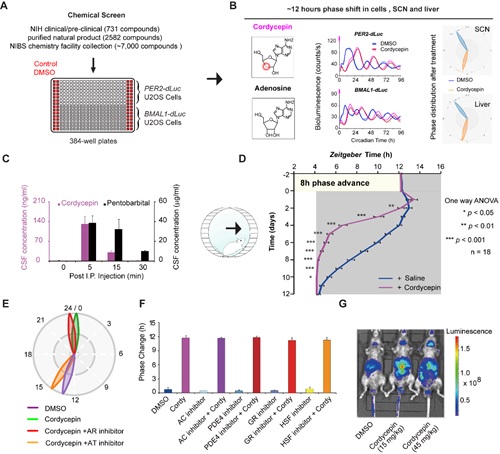
Figure 1. Chemical screen for circadian phase regulating molecules. (A) chemical screen strategy; (B) chemical structure of cordycepin and its antiphasic phenotype in culture cells and tissues; (c) cordycepin penetrates the BBB and enters CSF; (D) cordycepin shortens the time needed for an 8 h phase advance mouse jet leg assay; (E) cordycepin enters cells through an adenosine transporter and shifts the circadian phase; (F) inhibitors of known circadian synchronization pathways do not affect cordycepin’s phase shift ability; (G) cordycepin activates E-box gene transcription.
To identify the direct target of cordycepin, the authors applied a two level approach: given that the clock phenotype is likely involved in the regulation of the transcription of clock genes, the authors used siRNA libraries containing 1530 transcription factors and 463 epigenetic factors to evaluate which gene(s) may be functionally related to cordycepin-mediated Per2-dLuc perturbation. To identify proteins that physically interact with cordycepin, the authors conducted an IP-MS assay with U2OS cell lysates using functionally active biotinylated cordycepin which was provided by Dr. Xiangbing Qi's laboratory at NIBS. RuvB-like adenosine triphosphatase (ATPase) 2 (RUVBL2) was finally confirmed as the direct target through which cordycepin shifts the circadian phase.
RUVBL2 is a member of the AAA+ super-family of proteins, which participates multiple cellular processes by affecting ATPase activity, including for example chromatin remodeling TIP60 complex and the multi-subunit chaperone PAQosome complex. Interestingly, RUVBL2 has minimal ATPase activity, which is biochemically very similar to a simplest clock model: the KaiC oscillator in cyanobacteria. Whether RUVBL2 can function as the KaiC-like function in mammalian circadian system is currently under investigation as a priority of the authors. Cordycepin is immediately converted into cordycepin 5′-triphosphate (CoTP) after entering cells, and the phosphorylation of cordycepin is required for its phase-shift effect. When an adenosine kinase inhibitor was co-incubated with cordycepin, the phase shift effect was completely eliminated. A 2.50-Å structure of RUVBL2 in complex with CoTP and CoDP was obtained by Dr. Wei Zhang, supporting the known ATPase enzymatic activity of RUVBL2. The interaction between RUVBL2 and CoTP is functionally involved in the cordycepin-induced phase shift, which was confirmed in experiments that manipulated binding affinities (enhanced in D299Q mutant and weakened in A85T mutant) (Fig. 2).
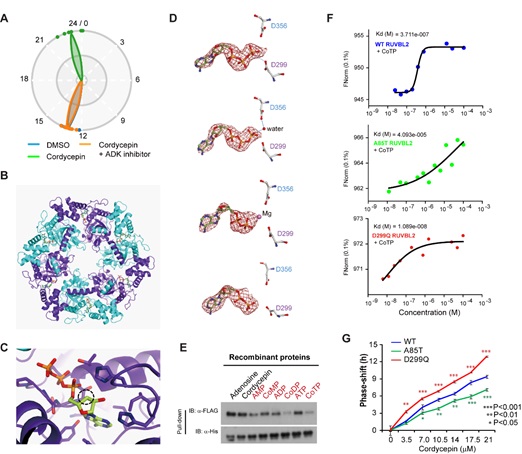
Figure 2. Interaction between CoTP and RUVBL2. (A) Adenosine kinase inhibitor prevents cordycepin phosphorylation and its phase phenotype; (B, C) protein structure of the CoTP/RUVBL2 interaction; (D) dynamic process of CoTP hydrolysis by RUVBL2; (E) CoTP and CoDP function to cause disassociation of the protein-protein interaction between RUVBL2 and BMAL1; (F) binding affinity between CoTP and the indicated RUVBL2 mutant variants; (G) phase shift ability of cordycepin in cells overexpressing the indicated RUVBL2 mutant variants.
A series of elaborately staged biochemistry experiments and mathematical modeling support that RUVBL2 is actually is a component of the core circadian clock, which itself is both regulated by the TTFL oscillator and participates with the TTFL complex to regulate circadian gene transcription. Upon binding with CoTP, the BMAL1/RUVBL2 interaction is disassociated, and the transcription status of the TTFL complex was converted from repression to activation, thereby to stimulating the transcription of E-box genes and reversing the circadian phase (Fig. 3). Consistently, cordycepin could not shift the circadian phase when the TTFL complex had already assumed its activated status (i.e., no phase effect when cordycepin was administered at the trough of E-box gene transcription).
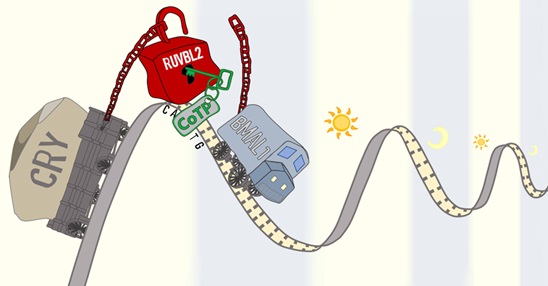
Figure 3. Working model of CoTP/RUVBL2 regulating the circadian phase. In mammals, the well-characterized circadian clock is a transcriptional/translational feedback loop oscillator. The transcription factors BMAL1/CLOCK bind to E/E′-box element in gene promoters, initiating transcription of genes including PER/CRY. When the translated PER/CRY proteins accumulate, they inhibit BMAL1/CLOCK transcriptional activity. These interactions together generate a delayed feedback loop that oscillates with an approximately 24-hour cycle. Here we found that E-box genes are repressed by a circadian regulatory super-complex comprising the previously known clock complex (CLOCK/BMAL1 and PER/CRY) and an RUVBL2-containing complex. When CoTP interferes with RUVBL2, this super-complex disassembles, and inhibitory CRY proteins leave the chromatin (whereas CLOCK/ BMAL1 remains on the chromatin); the lack of the CRY proteins relieves the repression, and E-box genes are transcribed. Subsequently, as the CoTP degradation and the super-complex begins to reassemble, a new clock oscillation starts (the cartoon sketch was prepared by Zhancong Xu and Ziqing Yu from the Zhang laboratory).
Starting from a chemical screen, this exciting study identified that cordycepin works intracellularly to cause very large phase shifts of circadian rhythms that could have broad translational relevance in humans. Moreover, this discovery of a CoTP/RUVBL2-gated circadian transcription mechanism substantially deepens the overall understanding of circadian clocks regulation.
Dr. Dapeng Ju from Dr. Erquan Zhang's laboratory and Dr. Wei from RPXDs (Suzhou) Co. Ltd. are the co-first authors of this work. Other contributing authors include Haijiao Zhao, Shuhua Ying, Meimai Liao, Zhancong Xu and Dr. Long Mei from Dr. Erquan Zhang's laboratory; Dr. Wei Li from RPXDs (Suzhou) Co. Ltd.; Jiawei Yan and Dr. Chao Tang from the Center for Quantitative Biology, Peking University; Dr. Xiaowen Xie and Dr. Luhua Lai from the School of Life Sciences, Peking University; Zhiqiang Wang, Guanshen Zhou, and Dr. Xiangbing Qi from the Chemical Center at NIBS; Dr. Jiawen Wang, Dr. Tao Cai, Dr. She Chen, Dr. Niu Huang, and Nannan Hou from the National Institute of Biological Sciences, Beijing; Dr. Joseph S. Takahashi and Dr. Noheon Park from the Department of Neuroscience, University of Texas Southwestern Medical Center. Dr. Erquan Zhang and Dr. Xiangbing Qi are the co-corresponding authors of this work. The study was supported by the Beijing Municipal Science & Technology Commission, MOST 973 grants, NNSFC, and Tsinghua University. The research work was carried out at National Institute of Biological Sciences, Beijing.
Note: This is the second research paper published by Science Translational Medicine from our institute. The previous research article is about developing an FTO inhibitor as a weight loss drug and its mechanism in regulating the metabolism of glucose and lipid, which was completed under a cooperation between Dr. Niu Huang's laboratory and Dr. Erquan Zhang's laboratory in our institute (and Yungui Yang laboratory at Beijing Institute of Genomics of the Chinese Academy of Sciences).
Original link:



